Your partner
in preventing infection.
Fast, safe, one-button
UV-C high-level disinfection for
ultrasound probes.
Our mission is to prevent healthcare-associated infections.
Healthcare-associated infections (HAIs) are a common and serious problem globally and locally. These infections are caused by cross-contamination of medical devices.
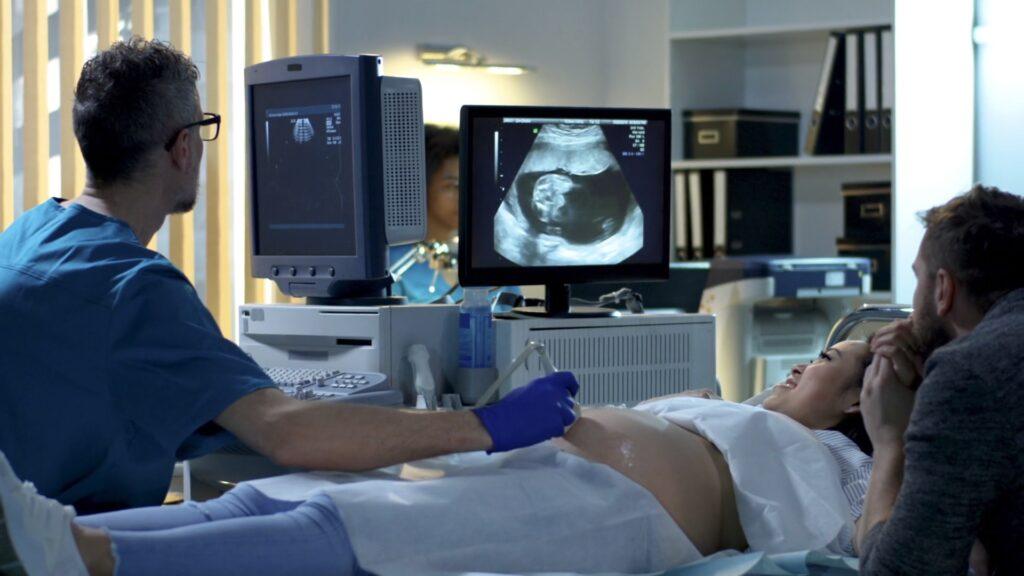
Women’s Health & IVF
13
%
of vaginal probes used with
sheaths test HPV positive after
disinfection with wipes.
Source: PLOS ONE
Cardiac Imaging
70
%
of HAIs can be prevented by implementing new infection
control interventions.
Source: WHO
ICU & Emergency
15
6
%
of patients in low- and mid-income countries will acquire at least one HAI during their hospital stay.
Source: WHO
Fast, safe, simple ultrasound probe disinfection
Yuvee®
Next-level
UV-C disinfection

Improve patient care and healthcare staff
safety while accelerating workflows.
Cutting-edge Innovation
Yuvee® technology is a leader in ultrafast, automated, high-level disinfection (HLD).
Trusted and recognised
We are globally recognised and known for excellence in scientific innovation.
Eco-conscious solutions
Our disinfection method doesn’t involve toxic chemicals — it’s safer for people and the planet.
Chemical-free sustainability

Get the latest news and information
Browse our articles, videos, and more to keep up with UV-C HLD news
Get the latest news
and information.
Browse articles, videos, and more to keep up with UV-C HLD news.

International Women’s Day: Working to Enhance Women’s Health

Germitec: UVC high-level disinfection technology for ultrasound probes
GE HealthCare Probe Compatible With Germitec UV-C HLD

Fertility Exhibition – January 2024

Unveiling Our New Website and Branding

